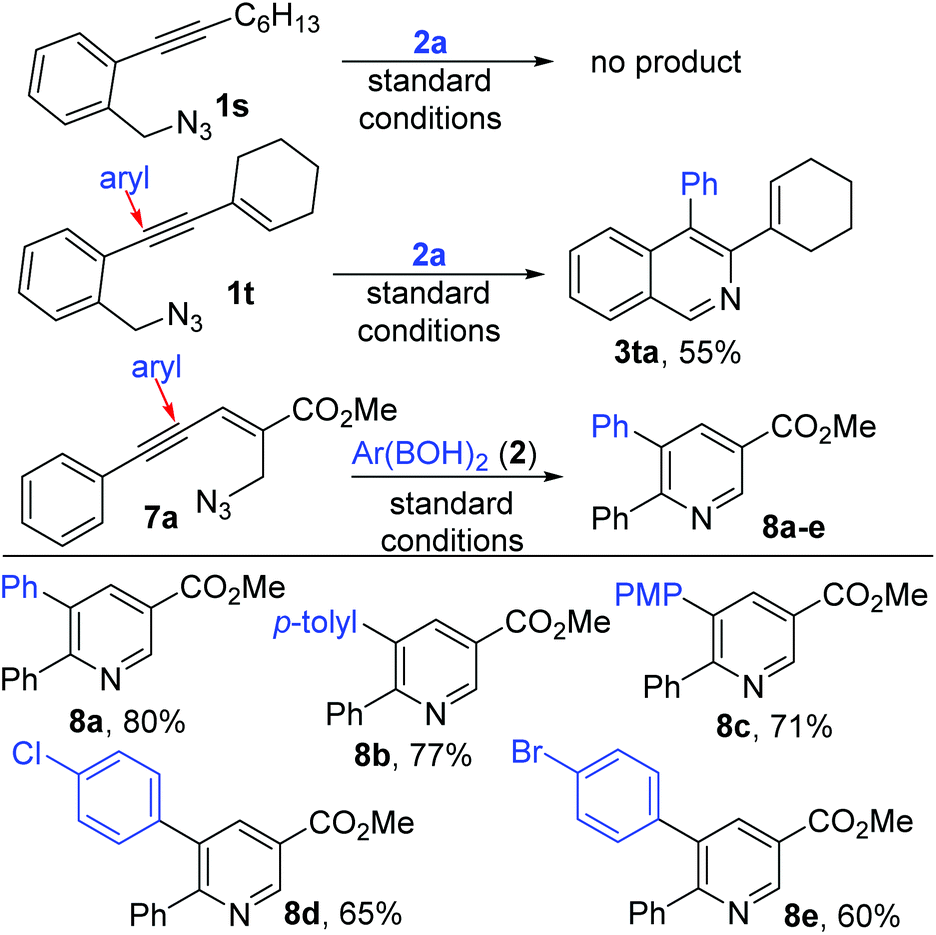
The reaction shows excellent functional group compatibility, affording functionalized diaryl ketones by selective NC cleavage. A one-pot preparation of anti-cholesterol drug fenofibrate (TriCor®) has been achieved on 10.0 mmol scale in 86 % yield. The reaction is promoted by a bench-stable, inexpensive Ni catalyst.
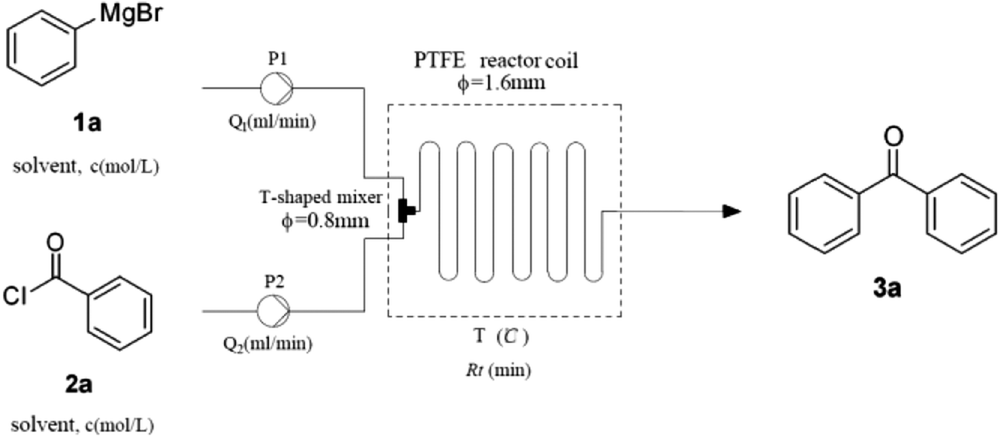
This method is suitable for rapid and large-scale synthesis of diaryl ketones. The dithiane arylation/hydrolysis can be performed in a one-pot procedure to yield a variety of diaryl ketones in good to excellent yields. The resulting 2,2-diaryl-1,3-dithianes were converted into diaryl ketones by either molecular iodine, N-bromo succinimide (NBS) or Selectfluor in the presence of water. In the presence of a Pd(NiXantphos)-based catalyst and aryl bromide, cross-coupling of the metallated 2-aryl-1,3-dithiane takes place under mild conditions (2 h at rt) with yields as high as 96 %. Ketone Synthesis by a Nickel-Catalyzed Dehydrogenative Cross-Coupling of Primary Alcohols. Use of MN(SiMe 3) 2 (M=Li, Na) base results in reversible deprotonation of the weakly acidic dithiane. This method entails the base-promoted, palladium-catalyzed direct C-H arylation of 2-aryl-1,3-dithianes with aryl bromides. An umpolung approach to the synthesis of diaryl ketones has been developed based on in situ generation of acyl anion equivalents and their catalytic arylation.
